This is an archive of information released in the past.
Disclaimer: It may contain broken links or outdated information. Some parts may not function in current web browsers.
*Visit https://humans-in-space.jaxa.jp/en/ for the latest information.

Experiment
- News
- Kibo Utilization Strategy
- Kibo Utilization Plan
- List of JAXA's Utilization Themes
- Experiment Facilities
- Space Environment Utilization
- Archive
Technological demonstration of protein crystallization at 4℃
JAXA has conducted protein crystallization (the JAXA PCG experiment) at 20℃ for a long period of time. Meanwhile, new devices to realize the experiment at 4℃ have been organized.
Crystallization at 4℃ realizes the crystallization of candidate drugs in high demand, such as unstable hydrosoluble proteins and membrane proteins. The first demonstration at 4℃ was conducted from April 10 - May 10 in the Japanese Experiment Module ("Kibo") whose samples were carried aboard the Dragon spacecraft (SpX-8) for both launch and return. For this demonstration mission, in addition to reference proteins that we prepared, researchers provided several protein samples.
Protein crystals returned from the International Space Station (ISS) were then transferred to Japan for immediate retrieval and viewing under a microscope on May 20 and 23, in order to grasp their condition. These obtained samples will be handed over to researchers for analysis at synchrotron radiation facilities (such as SPring-8).
Here we introduce some microscopic images of the JAXA-sponsored protein crystals. The results will be further studied, and we will strive to increase acceptable conditions for the JAXA PCG experiment.
Research theme: Analysis of high-resolution structures of protein kinase for the development of drugs
Principal Investigator: Kinoshita Takayoshi, Associate Professor at Osaka Prefecture University
【Overview】
Dr. Kinoshita's research group is studying the structure and drug development of protein kinase* that is an important target protein as pertaining to the development of molecularly-targeted drugs for rheumatism, cardiac enlargement, and cancers.
*Protein kinase: Protein-phosphorylating enzyme that involves the control of intracellular information (such as cell division and death)
It has been quite difficult to obtain the precise data of this research because when crystals of the target protein are grown on the ground, they tend to be clustered (where several crystals are combined together). Hence, the goal of the space experiment this time was to obtain single crystals so that precise 3D conformation could be determined.
The microscopic image below shows that the crystals grown in space are larger and that clusters are suppressed. These results will hopefully lead the acquisition of important knowledge about designing drugs for rheumatism, cardiac enlargement, and cancers.
【Remarks from Dr. Kinoshita】
1. Target of this crystallization experiment
Protein kinase that we study is the target protein of curative drugs for rheumatism and cardiac enlargement. Although we have studied its inhibitor complex, the positions of water molecules have yet to be identified.
When designing drugs, design based on a high-resolution structure including water molecules is very advantageous; therefore, we want to obtain high-quality crystals to realize the analysis of high-resolution structures by taking the opportunity afforded by a space experiment.
2. Feedback and expectations
Since 2011, we have sought to apply a space-crystallization experiment to protein kinase and repeated our proposals. However, at the ground test under the condition of 20℃, stable crystallization failed to reach that of a space experiment. Under 20℃, kinase proteins agglomerate and precipitate rapidly; therefore, we conduct experiments under 4℃ to suppress agglomeration.
JAXA now offers the opportunity for conducting a crystallization experiment at 4℃, thereby opening the door to space experiments involving unstable proteins. Although we have yet to conduct X-ray diffraction analysis, judging from the appearance, we consider the crystal to be of high quality and are satisfied with the experiment.
Crystals of protein kinase (© Osaka Prefecture University/JAXA) |
||
 |
 |
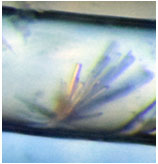 |
A crystal grown in space |
Clustered crystal grown on the ground (comparative experiment) |
|
*All times are Japan Standard Time (JST)
| Copyright 2007 Japan Aerospace Exploration Agency | Site Policy |